Hubbard Radio Washington DC, LLC. All rights reserved. This website is not intended for users located within the European Economic Area.
Hubbard Radio Washington DC, LLC. All rights reserved. This website is not intended for users located within the European Economic Area.
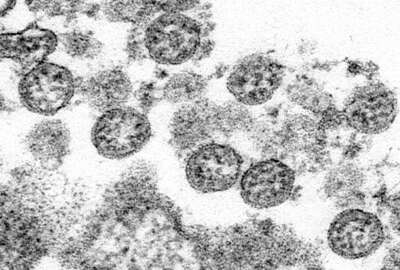
The Naval Research Lab used nanostructures to help discover how the coronavirus binds to and infects human cells. Now the National Center for Advancing Translat...
Best listening experience is on Chrome, Firefox or Safari. Subscribe to Federal Drive’s daily audio interviews on Apple Podcasts or PodcastOne.
A few weeks ago we brought you an interview with a Naval Research Lab scientist who used nano-structures to help discover how the coronavirus binds to and infects human cells. Now the other half of the story from the National Center for Advancing Translational Sciences. NCATS took the Navy-developed structures and did the tests on cells. With those details, Dr. Kirill Gorshkov joined Federal Drive with Tom Temin.
Interview transcript:
Tom Temin: Dr. Gorshkov, good to have you on.
Dr. Kirill Gorshkov: Thank you so much. Glad to be here.
Tom Temin: Tell us what you actually did because the purpose of this was to mimic these cells, the coronavirus or the COVID-19 cells, with the exterior structures that are believed to cause the binding. How did you discover how they worked?
Dr. Kirill Gorshkov: So after NRL confirmed that these nanoparticle quantum dot model viruses, we call them pseudo virions, but they can be thought of as a model virus that’s safe to use in a normal laboratory facility without the requirements of a biosafety level three or four containment facility for the actual deadly SARS CoV-2 virus. So after they confirm that this nanoparticle attaches to ACE2 the human receptor, that’s the first point of infection, they used purified ACE2 protein to confirm that this binding exists. They sent the nanoparticles over to NCATS, where I use human cells growing in micro-well plates. These are rectangular plates with clear bottoms and little raised wells that hold a small amount of liquid. The cells that we use, the human cells were designed to express the human ACE2 receptor attached to a green fluorescent protein. Now this allows us to see the protein and track where it is in the cell. And at the resting state, these ACE2 receptors in our human cells are at the cell surface, which is why the virus gets in so well because it just attaches to the surface of the cell. And so when I add the nanoparticles to cells, I can actually track the binding of the model virus to the cells, and subsequently the movement of both the model virus and the ACE2 receptor into the cell as a simulation of the infection process. So at NCATS, we can look at many different wells in a short amount of time. And we can capture images of the human cells that are exposed to the nanoparticles. And when we treat cells with drugs, in the effort to find a drug that can block SARS CoV-2 infection, we can make many different measurements of the microscope images to determine if the drug can block the model virus from attaching to and going inside of the cell.
Tom Temin: So in other words, you infected cells with not so much COVID-19, but with little glowing dots that had the COVID-19 binding mechanism on the outside? And how do you get a picture of something so tiny, I mean I think of a microscope is either an electron microscope, or an optical microscope, Which one did you have to use?
Dr. Kirill Gorshkov: So in this case, we do use optical microscopes. The specific type of microscope we use is a confocal microscope. And this allows us to magnify those cells in our micro-well plates quite well. And we use different sets of objectives. But essentially, the microscopes at NCATS are high throughput microscopes. So they’re these big boxes that sit on a benchtop. And they’re attached to a computer, we put the plate into the microscope, and then control it through the computer. And we set up a program that allows us to scan every single well of the plate in different positions in the wild very rapidly. And so we can take multiple colors of the same image using the different channels of the microscope. So that’s why we can see both the ACE2 receptor and these pseudo particles, these nanoparticle model viruses at the same time, and we can identify the cells as well. And so we use these big microscopes, really more like robots, to quickly scan across the plate. And this is the concept of high throughput, high content drug screening that we do at NCATS.
Tom Temin: And was there a moment in this experiment when you realized, hey, we are really onto something here and what was that like?
Dr. Kirill Gorshkov: It was a good feeling, because we knew that if I can see with my eyes, a change in the cells, when I add these quantum dots, I know that the computer can also identify these changes. And this is the concept of high content where we make many different measurements of an image, for example, the green color, the orange color from the quantum dots, the green color from the ACE2 receptor. And it was very exciting. And I knew we were onto something when I saw this green sheet on the cell surface turn into small dots, which is the internalization of the ACE2 receptor. And on top of these small dots was also the orange color, which was the quantum dot nanoparticle model virus. So because these two are together at the same position inside of the cell, I knew that these two were bound. And this was a good simulation of what we think is the end acidic mechanism for viral infection, it’s called endocytosis, when something from the exterior of the cellar at the cell surface is brought inside,
Tom Temin: This could lead them to the discovery of those drugs, or maybe there’s more than one, that can prevent to that cytosis in the first place.
Dr. Kirill Gorshkov: Yes, so we can either with our assay, specifically, we’re looking at the interaction between the viral spike protein that’s done on the surface of the virus that makes it look like this spiky ball and the ACE2 receptor. So with a drug, we can either block the spike ACE2 interaction, which would be great because the cell never even sees the SARS CoV-2 virus, or we can block the spike and ACE2 to partner from going inside of the cell. And that’s what our assay, our experiment, will allow us to see, drugs that target either of those processes.
Tom Temin: And where are we in that process?
Dr. Kirill Gorshkov: We are actually currently screening thousands of drugs and cats using these NRL developed nanoparticles. And we’re doing this in 1,536 well microplates. So these are very, very small plates, or the size of the plates is standard, but the wells inside of them are very, very small. And so this allows us to screen many different drugs within a single plate. And we have different drug libraries at endcaps that contain thousands of different drugs, both FDA approved, and also experimental drugs. And we are currently screening those libraries. We’re imaging those plates, we’re analyzing the images, and we are quantifying the differences between drug treatments and our control treatments. And so this hopefully, within the next couple of weeks, will lead to some interesting hits that we can follow up on and confirm using both the same as say that we’re currently doing the same experiment, we pick out those drugs and test them in different concentrations. But then we can also use other assays that we’ve developed at NCATS through orthogonal approaches. So alternative ways to measure this binding event. And we can also then cross check the compounds that we find the drugs that we find through the literature. So we can gather many different pieces of evidence, both experimentally and through the literature searching to really make sure that we’re doing our due diligence when we’re looking for these drugs. And so that is actively underway.
Tom Temin: And at some point then those drugs that you think have potential would have to go to human trials. And at that point with the same type of “cell,” I’m using that in quotes, developed by the NRL be used in humans safely.
Dr. Kirill Gorshkov: So the next steps after we confirm that the drugs that we find are working would have to be in advanced cell models. For example, right now, we’re working in 2D systems where the cells are just on a flat surface. But there are other ways to grow cells, and that is on top of each other. And so we can create these 3D models of different tissues, like the lungs, like the kidney, like the heart, and we can actually do experiments on those advanced 3D cell models. We can also move the drugs into animals. And there are several good animal models for SARS CoV-2 infection, and that would have to happen before we move into humans. Even if those drugs are FDA approved, there is still a preclinical development process that has to confirm these drugs are really working, and that they’re non toxic, that they’re safe, and not harmful to cells. So all of that has to happen in the preclinical space before the FDA can approve clinical trials. And at that point, the drugs would go through the phase one, phase two, phase three, safety and efficacy studies to make sure that the drugs are safe in humans. And then there would have to be some way to monitor the people that get the drugs to show that the people that are treated with the drug have a lower rate of infection by SARS CoV-2 than the people that receive a placebo. And that takes time. And that’s part of the reason why viral vaccine studies take a long time.
Tom Temin: Sure. And I guess we should know a little bit about you. How did you come to this type of work? How did you come to NCATS? Because you are not a 75 year old scientist that’s been at this for 50 years.
Dr. Kirill Gorshkov: That’s correct. I received my PhD from Johns Hopkins School of Medicine pharmacology and molecular sciences department, and that was in October of 2016. I immediately started a postdoctoral fellowship at the NIH within the National Center for Advancing Translational Sciences in the division of preclinical innovation and the biology group of the therapeutic development branch. And there I started converting and building upon my basic science training and my basic science foundation into more of a translational science research application. And I specifically wanted to search for drugs to help patients in need. And that’s where the high throughput screening training came in. And I built upon the fluorescence imaging that we used in this study that we’re talking about today. I got that training from Johns Hopkins. And so I’ve been combining my skill sets to develop and engage in drug discovery work. And so NCATS has a long running history of responding to pandemics, we did work on the Zika virus pandemic and the Ebola virus pandemic that started before I got there, but I was involved a little bit in it. And so the combined expertise of the things that I was doing before with the training that I got prior to SARS COV-2 pandemic, and the experience of the staff at the National Center for Advancing Translational Sciences created a nice environment for us to respond rapidly to the COVID-19 pandemic. And so I’m very happy to be a part of this work despite the terrible consequences of this virus. I think we’re doing an amazing job, and as much as we possibly can, at this time to search for drugs that can stop this disease.
Tom Temin: And you’re gonna stay on the case to the conclusion?
Dr. Kirill Gorshkov: Yes. This virus did not disappear in the summer, some people thought that it might because of the temperature change. Typically cold and flu season goes away in the summer, but this virus persisted. It’s quite infectious. And so we will definitely stay on the case as long as it takes. And we are trying right now to find small molecules and other therapeutic modalities to directly attack the virus and give patients some reprieve. There’s some drugs out there and the vaccines are definitely under development, and we can’t get those soon enough.
Tom Temin: Dr. Kirill Gorshkov is a scientist at the National Center for Advancing Translational Sciences, part of the NIH. We’re glad people like you are on the job and thanks for joining me.
Dr. Kirill Gorshkov: Thank you so much. It was a pleasure speaking with you today.
Copyright © 2024 Federal News Network. All rights reserved. This website is not intended for users located within the European Economic Area.
Tom Temin is host of the Federal Drive and has been providing insight on federal technology and management issues for more than 30 years.
Follow @tteminWFED
